
Our Translational Research Platform for Neurological Disease
SBH SCIENCESNeurological Disease Research Center
We partner with industry and academic institutions to support drug development across a broad range of neuroodegenerative disorders by providing comprehensive disease modeling state-of-the-art functional assays, and biomarker discovery and development services.
Our In-Vitro Models, Biomarker Platforms, and Functional Assays
We have developed a platform of functional assays in a growing range of in vitro models to support drug development programs for key indications in neurodegenerative disease drug development including:
- Cognitive-Dominant Neurodegenerative Disorders
- Movement-Dominant Neurodegenerative Disorders
- Motor Neuron Diseases
- Inflammatory Neurodegeneration
- Rare and Genetic Neurodegenerative Disorders

Functional Assays
Primary Neurons and Neuroblastoma Cells

Cell Viability and Neuroprotection

Mitochondrial Function

Oxidative Stress

a-Synuclein Aggregation and Clearance

Apoptosis and Cell Death Pathways

Target Engagement and MOA Validation

Neuronal Differentiation

Autophagy Lysosomal Pathway
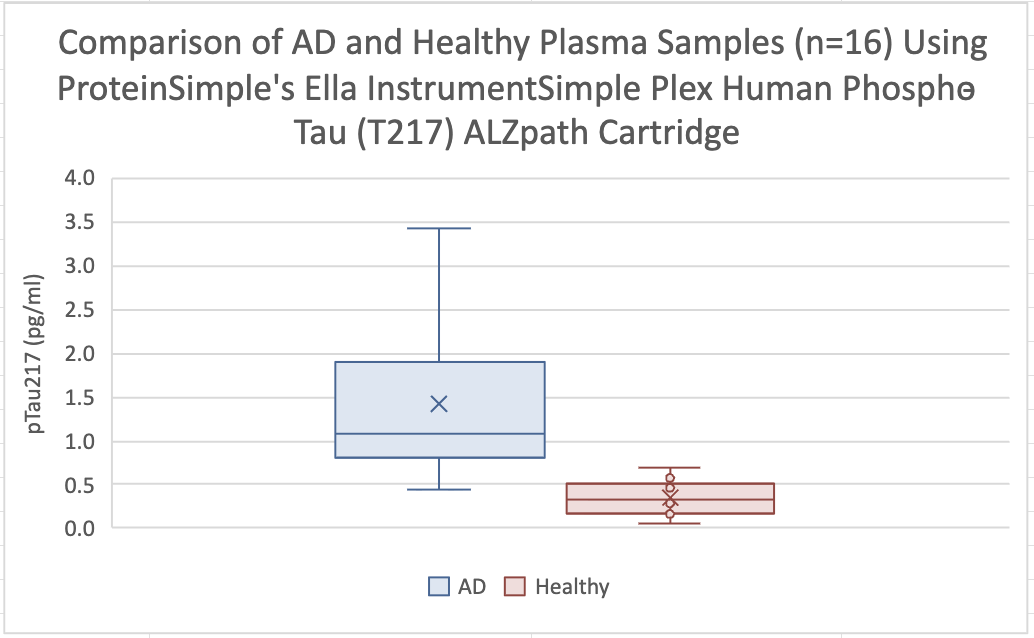
A New Way of Diagnosing Alzheimer's Disease
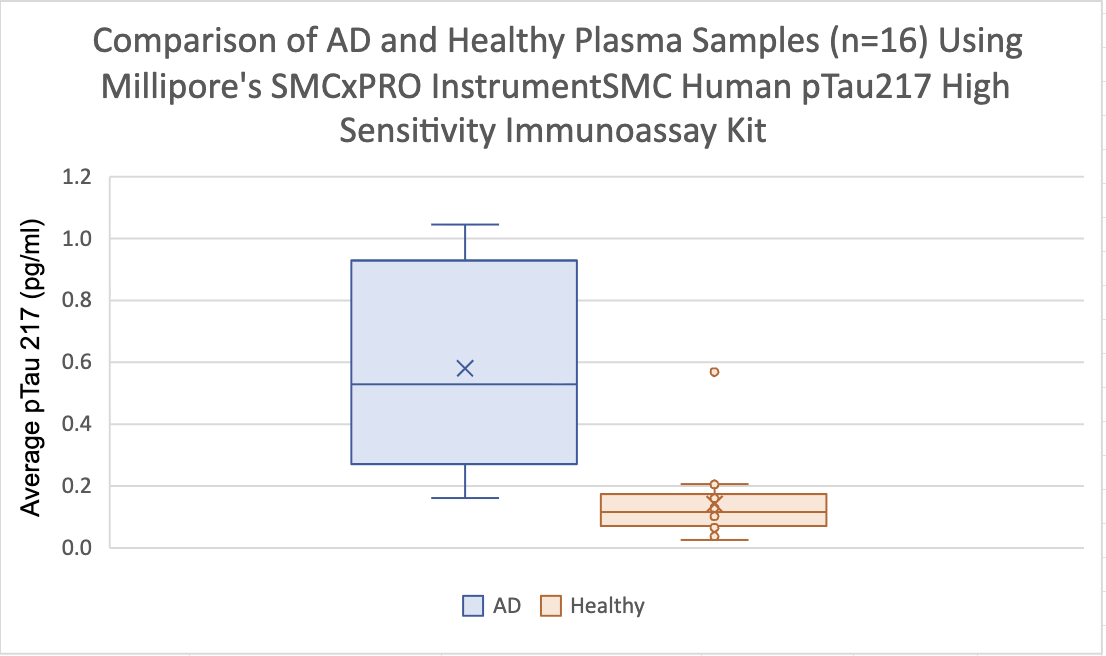
SMCxPRO
ELLA
We are currently validating the utilization of the ELLA for the analysis of β-Amyloid 1-40 and β-Amyloid 1-42 under CLIA. Shortly, all of these assays will be run under CLIA /GLP.

NEWNF-L Assay for Neurodegeneration Clinical Trials
Our validated and fully operational, CLIA & GCLP assay package measuring NF-L is immediately available for deployment in Phase I-III neurodegeneration trials, especially those studying Alzheimer's and ALS.
Biofluid Protein Biomarkers
| Biomarker | Indication | Biofluid |
|---|---|---|
| p-Tau217 |
Tau Phosphorylation (AD Specific) |
Plasma, CSF |
| p-Tau181 |
Tau Pathology |
Plasma, CSF |
|
p-Tau231 |
Early Tau Changes |
Plasma, CSF |
|
Aβ42 |
Amyloid Pathology |
CSF |
| Aβ42/40 Ratio |
Amyloid Burden |
Plasma, CSF |
|
Total Tau (t-Tau) |
Neuronal Injury |
CSF, Plasma |
|
GFAP |
Astrocytic Activatioon | Plasma, CSF |
|
NFL |
Axonal Degeneration | Plasma, CSF |
| Neurogranin | Synaptic Loss | CSF |
| SNAP-25 | Synaptic Dysfunction | CSF |
